
No matter your age, you can take steps each day to keep your blood pressure in a healthy range. What is the normal mmHg?Ī normal blood pressure level is less than 120/80 mmHg. 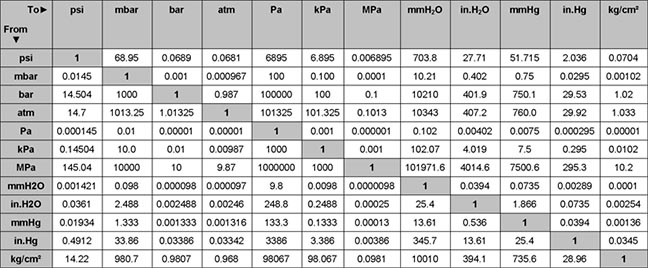
The readings are always given in pairs, with the upper (systolic) value first, followed by the lower (diastolic) value. How do you read mmHg?īlood pressure is measured in units of millimeters of mercury (mmHg). Often recommended during pregnancy to prevent edema. This is the range most often recommended for reducing swelling and preventing DVT (blood clots) during travel. … 15-20 mmHg: Also for minor and occasional swelling. MmHg stands for millimeters of mercury and it indicates the level of pressure or compression. If you need to convert from mmHg to kPa, multiply the number of mmHg by the conversion factor 0.133322387415.22 Related Questions Answers Found What is a good mmHg?Ī normal blood pressure level is less than 120/80 mmHg. To convert from kPa to mmHg, multiply the number of kPa by the conversion factor 7.500615758456564. What is the relationship between mmHg and kPa? The result will be your answer in atm.įor example, 1 atm multiplied by 76 equals 76 cmHg. To convert from atm (standard atmospheres) to cmHg (centimeters of mercury) multiply atm by 76. In countries where blood pressure is reported in kPa (kilopascals), multiply the kPa blood pressure reading by 7.500615758456564 to change the units. The reason why no conversion is required is that the blood pressure measurement is made in mmHg! 
In much of the world, you do not need to convert blood pressure values into mmHg. How do you convert blood pressure to mmHg? Other commonly used units of pressure and their abbreviations include pounds per square inch (psi), kilopascals (kPa), inches of water (inH2O), torr (Torr), technical atmosphere (kilograms-force per square cm kgf/cm2), barad (Ba), and bar (bar). In the case of water, you would need a column that is 10 meters high to measure one standard atmosphere! On the other hand, if you use a less dense liquid, the required height of the column is greater. Other applications include gas pressure readings, meteorology applications, or and in aviation so there is a limit on the height of any instrument.įor example, to measure one standard atmosphere of pressure (atm), you only need a column that is 760 mm tall (0.76 meters) if you use mercury as the liquid. A pressure column, also called a manometer, is often used inside a laboratory. A dense liquid is helpful for pressure column applications because it minimizes the height needed for a column, which is an important practical concern. Historically, pressure measurements were made in millimeters of mercury (mmHg) because mercury is the densest liquid at room temperature. As outside air pressure increases or decreases, the change in the weight of the pressure on the column of mercury will cause mmHg to rise or fall. In meteorology, millimeters of mercury (mmHg) is used in weather forecasts and reporting. Mmhg is a manometric unit of pressure equal to the pressure created by a column of mercury that is one millimeter high. There are 760 mmHg in a standard atmosphere but only 0.0013157894736842 standard atmospheres (atm) in one mmHg. Yes, one standard atmosphere (atm) is greater than one millimeter of mercury (mmHg). Yes, 1 standard atmosphere (atm) is exactly equal to 760 millimeters of mercury (mmHg). Both the multiplication and division-based approaches will give you the correct solution. The second option to change the units is to divide atm by the conversion factor of 0.0013157894736842. The first option is to multiply atm by the conversion factor of 760, which is the number of atm in 1 mmHg. You have two conversion factors to choose from to convert standard atmospheres (atm) to millimeters of mercury (mmHg). You will most frequently encounter mmHg from blood pressure readings and a meteorologist’s weather reports in everyday life. It is defined as equal to the pressure exerted by a column of mercury that is one millimeter high under standard gravity. What does mmHg stand for?Ī mmHg is a manometric unit of pressure that stands for ‘millimeters of mercury’. It is equivalent to the average atmospheric pressure on Earth at sea level. A Standard atmosphere (atm) is a unit of pressure defined as 101,325 Pa, 760 mmHg, 1.01325 bar, 760 torr, 4.6959 psi, or 1 ata (atmosphere absolute).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
